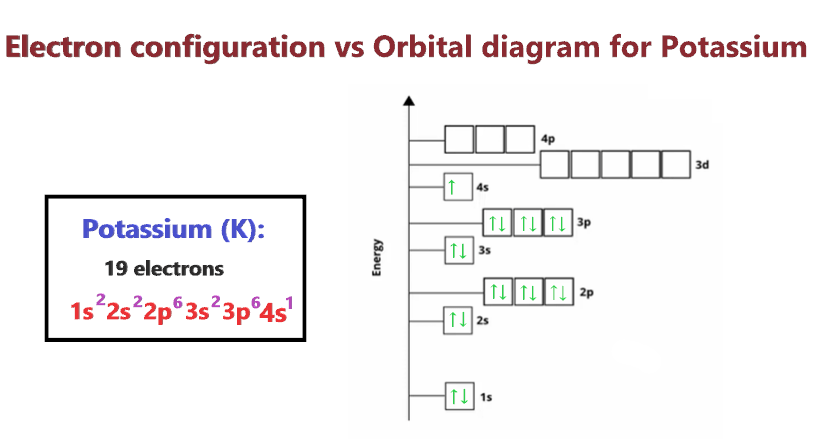
The third energy level has one electron in the s-orbital. The second energy level has eight electrons split between the s and p-orbitals. The orbital diagram for potassium indicates that the first energy level has two electrons in the s-orbital. Electron configuration 4s 1: Electrons per shell: 2, 8, 8, 1: Physical properties Phase at STP: solid: Melting point: 336.7 K (63.5 ☌, 146.3 ☏) Boiling point: 1030.793 K (757.643 ☌, 1395.757 ☏) Density (near r.t. It is a visual representation that uses boxes and arrows to show the energy level and number of electrons in each orbital. To write the orbital diagram for the Potassium atom (K) first we need to write the electron configuration for just K. 
The remainder of potassium's electron configuration is given by the 4s 1 electron.Īn orbital diagram is another way to represent the electron configuration of an atom. The symbol indicates that the first two energy levels of potassium have the same electron configuration as argon. Argon is a noble gas that has a full outermost energy level configuration, which in this case is the third energy level. The noble gas notation of potassium is 4s 1. It uses the symbol of the noble gas element that is located in the previous energy level followed by the electron configuration of the remaining electrons. Noble gas notation is a shorthand method of representing the electron configuration. This means that the first energy level has two electrons, the second has eight electrons, and the third energy level has one electron. The full electron configuration of potassium can be listed as 1s 2 2s 2 2p 6 3s 2 3p 6 4s 1. electron ground - state configuration of potassium is found, experimentally, to be 1s2 2s2 2p 3s2 3p6 4s1 this is interpreted to mean that, in many. The electron configuration describes the arrangement of electrons in different energy levels in an atom. This outermost shell is known as the valence shell, and the electrons trapped inside are known as valence electrons.Potassium has an atomic number of 19 which means that each potassium atom has 19 protons and 19 electrons. The number of electrons in the outermost shell of a given atom determines either its reactivity, or a propensity to form chemical bonds with other atoms. The electrical arrangement refers to the distribution of electrons in various shells / orbits / levels of energy at each atom. Atoms in individual elements have various electrical properties, based on the atomic number of each element. Note- The outermost shell of any element is the shell of Valence. Therefore, if electrons are to be removed or added, they get removed from or added to the shell with the highest value of n. Hence, the electronic configuration of potassium is 2,8,8,1 and not 2, 8, 9. Therefore, according to the law of the octet, an atom's outermost shell can accommodate up to 8 electrons (with the exception of a K shell that can accommodate up to 2 electrons). The elements that follow this law usually include the s-block elements and the p-block elements (except hydrogen, helium, and lithium). The halogens, oxygen, nitrogen, and carbon molecules are believed to follow the laws of the octet. The law of the octet applies only to the principal group elements. It is based on the fact that the atoms of the main group elements appear to be involved in chemical bonding in such a way that each atom of the resulting molecule has eight electrons in the shell of valence. Octet law: The octet rule states that atoms are most stable when eight electrons occupy their valence shells. Hint: In order to deal with this question first we will elaborate the octet rule further we will take an example of potassium to prove the question statement is true.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
